Ultrasonography of the reticulum, rumen, omasum and abomasum before, during and after ingestion of hay and grass silage in 10 calves
Braun, U., Gautschi, A., Tschuor A., Hassig, M.
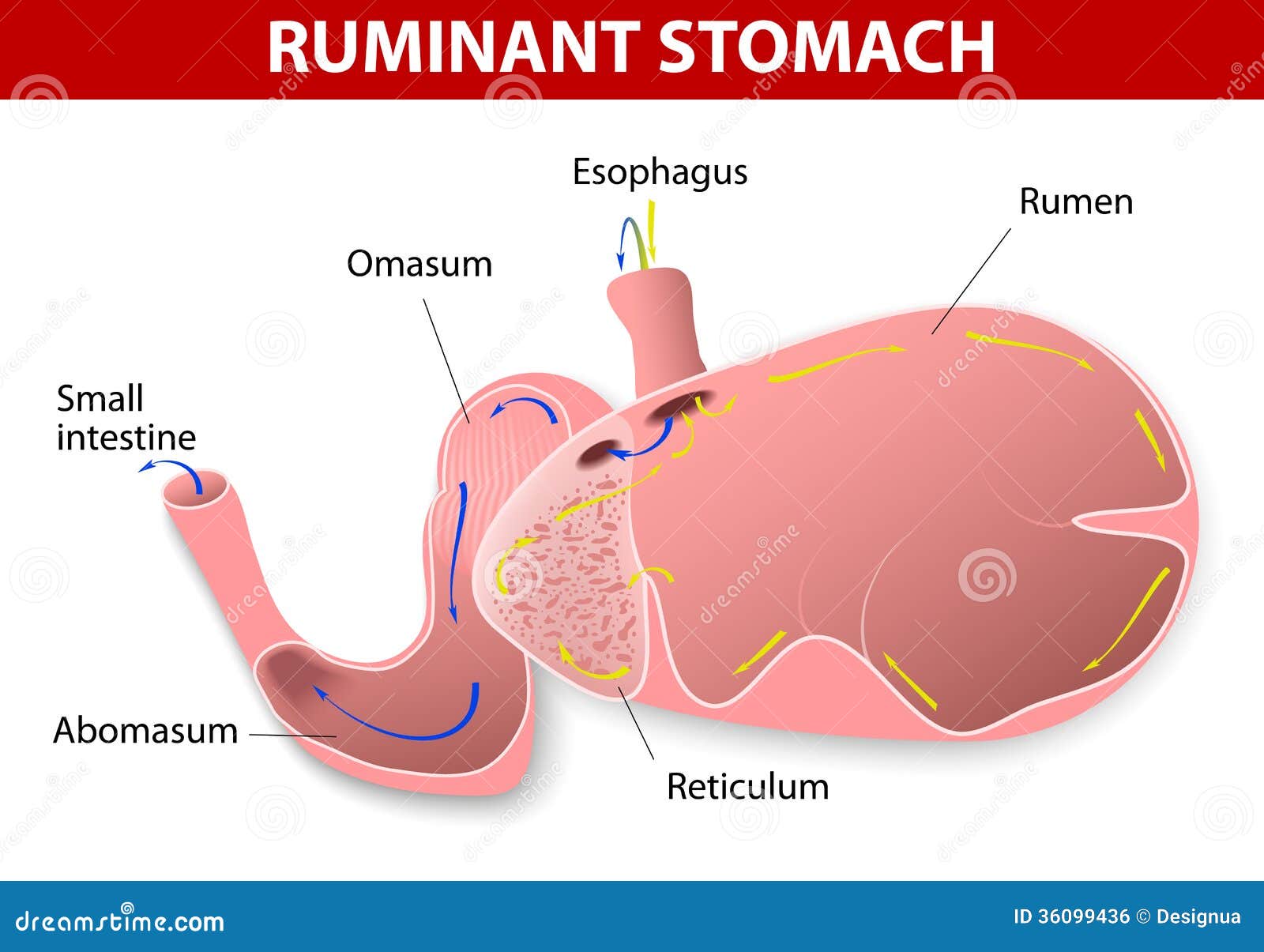
Introduction
Ultrasonography is a diagnostic medical procedure that uses high frequency sound (ultrasound) waves to produce images of internal organs and other tissues. It is especially useful in the evaluation of many diseases that affect the bovine digestive tract (Braun et al. 2012). The ultrasonographic findings of the rumen, reticulum, omasum and abomasum of healthy adult cows have well documented. The ultrasonographic images of the stomach compartments of milk-fed calves have also been documented. However, the results of both these studies cannot necessarily applied to calves that have been fed roughages like hay. This is because the relationship between the size of the forestomachs and abomasum in calves is different to that of adult cattle. Also, the physiological changes that occur in the digestive tracts of calves that consume milk is different from that of those calves that consume high fibre diet (Braun et al, 2012). In milk-fed calves, digestion occurs mainly in the abomasum while in hay-fed calves, digestion occurs mainly in the rumen. The goal of this study was to investigate the ultrasonographic appearance of the rumen, reticulum, omasum and abomasum in ruminating calves before, during and after being fed hay.
Results
Ultrasonagraphy of the Reticulum
The reticulum could be visualised in all calves before feeding. The appearance of the reticular wall as well as contractions were similar to those in adult cattle. The ultrasonographic appearance of the reticulum was the same before, during and after feeding. The number of contractions during feeding was significantly larger than the number recorded before and after feeding.
Fig 1: Ultrasonogram of the reticulum of a 3-month-old Simmental calf taken from the left ventral paramedian region using a 5.0 MHz linear transducer. (1) Ventral abdomen, (2) Spleen, (3) Reticulum, (4) Cartilage part of the rib, Cr Cranial, Cd Caudal
Ultrasonography of the Rumen
On the left side the rumen could be seen from 7th to 12th intercostal space and the entire flank region in all the calves. The visible size of the rumen was largest at 38.6±3.7 cm in the cranial flank. Its visible size decreased cranially because of superimposition of the lungs. In all calves a gas cap was seen in the dorsal sac of the rumen, which caused a reverberation artifact running parallel to the ruminal wall. The transition from gas to ingesta was recognised by an abrupt end of the reverberation artifacts lines. The rumen varied in size from 7.5-14.0 cm. There was no difference in the ultrasonographic appearance of the rumen before, during and after feeding.
Fig2: Ultrasonogram of the dorsal sac of the rumen in a 3-month-old Brown Swiss calf viewed from the 11th intercostal space on the left side and in the region of the dorsal gas cap using a 5.0 MHz linear transducer. (1) Lateral abdominal wall, (2) Rumen wall, (3) Reverberation artifact at the levof the dorsal gas cap in the rumen, Ds Dorsal, Vt Ventral
Ultrasonography of the omasum
The omasal appearance was the same before, during and after feeding in all of the calves. The omasum was largest in the 8th intercostal space at 8.5±2.2 cm and became progressively smaller towards cranial ad caudal points. Only the omasal wall and leaves closest to the transducer were visible. Omasal motility was not detected in any of the calves during the observation period
Fig. 3: Ultrasonogram of the omasum of a 3-month-old Simmental calf viewed from the 8th intercostal space on the right side using a 5.0 MHz linear transducer. (1) Lateral abdominal wall, (2) Liver, (3) Omasum, Ds Dorsal, Vt Ventral
Ultrasonography of the Abomasum
The abomasum was visible in all of the calves to the left and right of the ventral midline before feeding. It lay immediately adjacent to the abdominal wall in the region of the ventral midline. The abomasal walls were clearly seen in all the calves and appeared homogenous in seven calves and heterogeneous in three calves. The pylorus was also seen. The ultrasonographic appearance was the same before and after feeding.
Fig 4: Ultrasonogram of the abomasum of a 3-month-old Simmental calf viewed from the left ventral paramedian region using a 5.0 MHz linear transducer. (1) Ventral abdominal wall, (2) Abomasum, (3) Reticulum, (4) Cranial dorsal blind sac of the rumen, (5) Ventral sac of the rumen, Cr Cranial, Cd Caudal
Discussion
The results for the ultrasonography of the reticulum showed that the reticulum of calves can be visualized like that of the adult cattle. This is in contrast to milk-fed calves in which the reticulum could not be seen because it was too small and not in contact with the abdominal wall. The frequency of the reticular contractions before feeding was almost identical to the frequency reported in resting cows but slightly higher than in milk-fed calves.
The rumen was readily seen in all calves, although compared with adult calves, it was very small. The rumen was not seen in newborn calves, presumably because of its small size. The transition from gas to ingesta was seen in all calves. Differentiation is not seen milk-fed calves because their diet consisted exclusively of milk. In contrast to calves, differentiation of the ingesta and ventral fluid phases has been described in cows.
The omasum was seen in all ruminating calves, similar to cows. The omasum in the calves was considerably smaller than those in cows but larger than of the milk-fed cows.
The abomasum was found in the same location in the calves in this study as well as in milk-fed calves. An increase in the abomasum was seen in calves fed milk immediately after ingestion of milk, but this was not seen in the calves fed hay. This is because the esophageal groove transported milk directly into the abomasum, whereas in claves eating roughage, the feed passes into the rumen first and only small portins are passed into the abomasum.
Critique
The goals and findings of this study presented by Braun at al. were easy to understand and the results were organized in clear and concise manner and are easily understood. The authors did a good job in addressing the objective of the study. I liked the paper as it not overly long and the methods were easy to follow with not very many “big” words used. The results of the experiment support the authors’ claims that even though ultrasonography of stomach compartments in adult cattle and milk-fed calves have been documented, they cannot be used to estimate the stomach compartments in ruminal calves.
The problem I found with this paper was that the sample size was really small. They used only 10 calves to estimate the appearance and features of rumen, reticulum, omasum and abomasum of all calves. Also, only two breeds of cattle were used and the stomach compartments could be different in breeds of cattle.
Although the authors wrote that the study protocol was approved by the Animal Care Committee of the Canton of Zurich, Switzerland, I did not like that the calves had to fast for 10 hours before each of the experiments that took place over three days so the calves fasted ten hours on each of the three days.
Another problem I found with the study is that it took place over three days and on day 2 was when ultrasonography of the stomach during feeding was done. As the calves were fasted for 10 hours before they were fed for an hour during which the ultrasonographic images were taken, I find that the calves may not have been able to eat as well as they could have and this could slow their metabolism. Also, the after feeding reading was done the next after another 10 hours fasting and feeding for an hour. The ultrasonographic readings were taken 2 hours after feeding which I think is a short time to determine the abomasal and omasal content as the ingesta is probably still in the rumen and reticulum and have to be regurgitated several times before it moves to the omasum and abomasum. Another problem that this paper had that the images were not exactly the best as they are grainy and not very clear (as seen above).
Overall, the authors did a good job in addressing the objectives of the study, although more work could be done by using a larger sample size as well as more breeds of cattle.
References
Braun, U., Gautschi, A., Tschuor A., Hassig, M. (2012). Ultrasonography of the reticulum, rumen, omasum and abomasum before, during and after ingestion of hay and grass silage in 10 calves. Research inVeterinary Science, 93(3), 1407-1412. doi: http://dx.doi.org.qe2a-proxy.mun.ca/10.1016/j.rvsc.2012.03.012
This paper can be found here